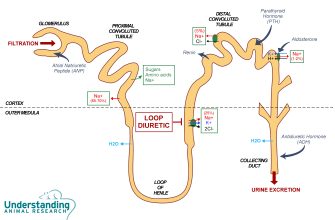
Focus on securing intellectual property rights immediately. Canadian patent law offers robust protection, but navigating the system requires proactive strategies. File promptly; delays can significantly impact your market position. Consider a comprehensive patent search to ensure your innovation is truly unique.
Secure early-stage funding through government grants and venture capital. Explore programs like the Industrial Research Assistance Program (IRAP) for direct support and mentorship. Prepare a compelling business plan highlighting the market potential and your team’s expertise to attract investors.
Build strong relationships with Health Canada early in the development process. Understanding their regulations and approval pathways is paramount. Proactive engagement minimizes delays and maximizes the chances of a successful market entry. This includes thorough documentation and compliance with Good Manufacturing Practices (GMP).
Remember: Strategic partnerships are key to accelerating growth. Collaborate with experienced players in the Canadian pharmaceutical industry for distribution, marketing, and clinical trials. Leverage their established networks and infrastructure to rapidly expand your market reach.
Key takeaway: A well-defined commercialization strategy, incorporating intellectual property protection, funding acquisition, regulatory compliance, and strategic alliances, will significantly increase the chances of success for your greenfield drug venture in Canada.
- Canadian Drug of Greenfield: A Detailed Overview
- Regulatory Compliance: Navigating the Canadian Drug Approval Process
- Market Access and Reimbursement Strategies: Securing Market Share
- Funding and Investment: Securing Capital for Greenfield Projects
- Supply Chain Management and Manufacturing: Ensuring Reliable Production
- Post-Market Surveillance and Commercialization: Maintaining Market Position
- Defining “Greenfield” in the Canadian Pharmaceutical Context
- Challenges Faced by Greenfield Pharmaceutical Companies in Canada
- Funding and Financial Management
- Talent Acquisition and Retention
- Government Regulations and Approvals for Greenfield Drug Development
- Pre-Clinical Testing and Investigational New Drug (IND) Application
- Clinical Trials and Regulatory Interactions
- New Drug Submission (NDS)
- Post-Market Surveillance
- Key Considerations
- Intellectual Property Protection
- Funding and Investment Opportunities for Greenfield Pharmaceutical Ventures
- Strategic Partnerships
- Securing Series A Funding
- Public Offerings (IPO)
- Funding Sources Comparison
- Beyond Funding: Intellectual Property Protection
- Success Stories and Case Studies of Greenfield Drug Development in Canada
- The Future of Greenfield Pharmaceuticals in the Canadian Market
- Strategic Partnerships and Acquisitions
- Regulatory Navigating & Market Access
- Investing in Digital Health Technologies
Canadian Drug of Greenfield: A Detailed Overview
Greenfield projects in the Canadian pharmaceutical industry present unique opportunities and challenges. Focus on securing robust intellectual property protection early. This minimizes risks associated with patent infringement and ensures market exclusivity.
Regulatory Compliance: Navigating the Canadian Drug Approval Process
Understand Health Canada’s rigorous drug approval process. Thorough pre-clinical testing and clinical trials are mandatory, requiring significant resources and meticulous documentation. Engage experienced regulatory consultants to ensure compliance and expedite the approval timeline. Complying with Good Manufacturing Practices (GMP) is paramount for production facilities.
Market Access and Reimbursement Strategies: Securing Market Share
Develop a targeted market access strategy. This involves understanding provincial drug formularies and negotiating favorable reimbursement rates with public and private payers. Pricing strategies should reflect the drug’s value proposition and competitive landscape. Building strong relationships with key opinion leaders and healthcare providers is also vital for market penetration.
Funding and Investment: Securing Capital for Greenfield Projects
Secure adequate funding through a combination of sources, including venture capital, government grants, and private equity. Develop a compelling business plan highlighting the drug’s potential, market size, and return on investment. Clearly demonstrate a strong management team and a robust intellectual property portfolio to attract investors.
Supply Chain Management and Manufacturing: Ensuring Reliable Production
Establish a reliable and efficient supply chain. This involves securing high-quality raw materials, establishing manufacturing partnerships, and implementing robust quality control measures. Consider various manufacturing options, including contract manufacturing or establishing your own production facilities, based on budget and scale of operations.
Post-Market Surveillance and Commercialization: Maintaining Market Position
Implement a comprehensive post-market surveillance program to monitor drug safety and efficacy. Actively collect and analyze data to identify potential safety concerns and address them promptly. Build a strong commercialization strategy encompassing sales, marketing, and distribution channels to maximize market reach and penetration.
Defining “Greenfield” in the Canadian Pharmaceutical Context
In Canadian pharmaceuticals, “greenfield” signifies a completely new pharmaceutical operation, built from the ground up. This differs from a brownfield project, which involves expanding or repurposing an existing facility. A greenfield project might involve constructing a new manufacturing plant, establishing a research and development centre, or creating a wholly new distribution network.
Consider the regulatory hurdles: Navigating Health Canada’s approvals for new facilities and production processes is a significant undertaking, requiring meticulous planning and substantial investment. Successful projects proactively address these challenges early.
Funding sources often include private investment, government grants (such as those focused on innovation or regional development), and potentially strategic partnerships with established players. Secure funding before commencing construction is critical.
Location selection impacts logistical costs and access to skilled labor. Proximity to universities with relevant research programs and a skilled workforce is highly beneficial. Factors such as available infrastructure, transportation links, and proximity to potential markets should be carefully weighed.
Successful greenfield projects in the Canadian pharmaceutical sector prioritize clear objectives, robust planning, and strong regulatory compliance. This systematic approach minimizes risks and maximizes the chances of a successful venture.
Challenges Faced by Greenfield Pharmaceutical Companies in Canada
Secure robust intellectual property (IP) protection early. Canadian patent laws are complex; proactive legal counsel is paramount to safeguarding your innovations. Expect significant legal fees, but consider this a vital investment.
Navigate Canada’s stringent regulatory environment effectively. Health Canada’s approval processes are rigorous, requiring extensive documentation and clinical trials. Budget substantial time and resources for regulatory compliance, aiming for proactive engagement with Health Canada throughout the process.
Funding and Financial Management
Securing sufficient seed funding and Series A financing is challenging. Canadian venture capital investment in pharmaceuticals can be competitive. Develop a compelling business plan highlighting your unique selling proposition and market potential. Explore government grants and incentives to supplement private investment.
Manage operational costs efficiently. Rent, salaries, and research expenses can quickly drain resources. Implement lean operational strategies from the outset, prioritizing strategic resource allocation and potentially considering outsourcing non-core functions.
Talent Acquisition and Retention
Attracting and retaining skilled pharmaceutical professionals is difficult. Canada’s pharmaceutical industry competes for a limited pool of talent. Offer competitive salaries and benefits packages, creating a positive and engaging work environment to maintain employee loyalty.
Establish strong partnerships with academic institutions and research centers. Collaboration can provide access to specialized expertise and accelerate drug development. Consider co-op programs and internships to cultivate future talent pools.
Government Regulations and Approvals for Greenfield Drug Development
Secure Health Canada approval early. This involves navigating several phases, beginning with pre-clinical testing and culminating in a New Drug Submission (NDS).
Pre-Clinical Testing and Investigational New Drug (IND) Application
- Conduct rigorous pre-clinical studies demonstrating safety and efficacy. These must adhere to Good Laboratory Practice (GLP) guidelines.
- Prepare a detailed IND application, outlining your study protocols, data, and manufacturing processes.
- Engage with Health Canada early – informal meetings can clarify expectations and expedite the review process.
Remember, robust data significantly increases your chances of approval.
Clinical Trials and Regulatory Interactions
- Design clinical trials aligning with ICH guidelines. Clear protocols are vital.
- Maintain ongoing communication with Health Canada throughout the clinical trial phases, addressing any queries promptly.
- Ensure your manufacturing processes meet Good Manufacturing Practice (GMP) standards.
- Submit interim reports and address any concerns raised by Health Canada’s reviewers.
New Drug Submission (NDS)
Your NDS is the comprehensive package submitted for final approval. This requires meticulous preparation.
Post-Market Surveillance
- Post-market surveillance is ongoing, requiring vigilance in monitoring drug safety and efficacy.
- Report adverse events promptly to Health Canada.
- Continuously update Health Canada on any changes to your manufacturing or labeling.
Key Considerations
Intellectual Property Protection
- Secure patents to protect your intellectual property throughout the development process.
Proactive engagement with Health Canada streamlines the approval process, reducing time and resources expended.
Funding and Investment Opportunities for Greenfield Pharmaceutical Ventures
Secure seed funding through government grants like those offered by the Canadian Institutes of Health Research (CIHR) or provincial agencies. Explore angel investors specializing in life sciences; many actively seek early-stage pharmaceutical companies. Venture capital firms frequently invest in promising biotech startups; research firms focused on healthcare investment are a great resource to identify them.
Strategic Partnerships
Collaborate with established pharmaceutical companies for joint ventures or licensing agreements. This provides access to their resources and expertise, reducing the financial burden of independent development. Seek partnerships with research institutions for technology transfer and collaborative research projects. This lowers R&D costs and enhances credibility.
Securing Series A Funding
After demonstrating proof of concept, target Series A funding from venture capital or private equity firms. A compelling business plan highlighting market potential, intellectual property, and a strong management team is crucial. Prepare a detailed financial projection, including projected revenue streams and milestones.
Public Offerings (IPO)
Once you’ve achieved significant milestones and market traction, consider an Initial Public Offering (IPO) to raise capital. This requires substantial regulatory compliance, audited financials, and a comprehensive investor relations strategy. Consider hiring experienced investment banking firms to guide you through the process.
Funding Sources Comparison
| Funding Source | Pros | Cons |
|---|---|---|
| Government Grants | Lower risk, potentially non-dilutive | Competitive application process, stringent requirements |
| Angel Investors | Early-stage investment, mentorship | Limited capital compared to VCs |
| Venture Capital | Significant capital injection, industry expertise | Dilutive, high expectations for returns |
| Strategic Partnerships | Resource access, reduced risk | Potential loss of control, profit sharing |
| IPO | Large capital raise, enhanced credibility | High regulatory burden, significant cost |
Beyond Funding: Intellectual Property Protection
Aggressively protect your intellectual property through patents and trade secrets. This significantly increases your company’s value and attracts investors.
Success Stories and Case Studies of Greenfield Drug Development in Canada
Consider Biolyse Pharma’s successful development and launch of its generic drug portfolio. Their strategic focus on complex generics resulted in significant market share gains and substantial revenue growth within five years. This demonstrates the potential for Canadian companies to compete globally in the pharmaceutical sector.
Another example is Apotex Inc.’s consistent track record of bringing innovative generic medications to market. Their agile development processes, coupled with strong regulatory expertise, allowed them to reduce time-to-market significantly, improving profitability and patient access to affordable drugs. Their investment in research and development into complex generic formulations continues to yield positive results.
Smaller biotech firms like Precision Biologics have shown the value of niche drug development. By focusing on unmet medical needs within specific therapeutic areas, they secured strategic partnerships and substantial funding, enabling clinical trial progression and ultimately, product commercialization. This highlights the advantages of a targeted approach.
Furthermore, several Canadian universities are actively involved in translational research, facilitating the transfer of research findings into new drug development programs. McGill University’s strong collaboration with industry partners has resulted in several promising drug candidates advancing into clinical trials. This demonstrates the critical role academic institutions play in the Canadian drug development ecosystem.
These examples illustrate that successful greenfield drug development in Canada requires a combination of strategic planning, robust research, effective regulatory navigation, and strategic partnerships. A focus on innovation, coupled with a clear understanding of market demands, is crucial for achieving commercial success.
The Future of Greenfield Pharmaceuticals in the Canadian Market
Canadian Greenfield pharmaceutical companies should aggressively pursue niche therapeutic areas with limited competition. Focus on developing innovative, first-in-class drugs addressing unmet medical needs within geriatrics and rare diseases, leveraging Canada’s strong research institutions for accelerated development. This targeted approach minimizes risk and maximizes return on investment.
Strategic Partnerships and Acquisitions
Strategic collaborations with established Canadian or international pharmaceutical companies offer access to crucial resources like manufacturing and distribution networks. Acquiring smaller biotech firms with promising drug pipelines provides immediate market access and expands intellectual property portfolios. Successful partnerships should center around complementary expertise and shared market goals, avoiding conflicts of interest. Consider focusing on companies with a strong digital presence, accelerating product launch and marketing.
Regulatory Navigating & Market Access
Proactive engagement with Health Canada is critical. A thorough understanding of the regulatory pathway, including streamlined submission processes, accelerates drug approvals. Collaborate with Health Canada throughout the development process to avoid delays. Develop robust market access strategies, negotiating favorable reimbursement terms with provincial healthcare payers early, ensuring timely patient access to new therapies. This will improve the speed of revenue generation and long-term sustainability.
Investing in Digital Health Technologies
Integrating digital health technologies into product development and marketing is paramount. Leveraging AI for drug discovery, precision medicine, and personalized treatment plans enhances efficiency and effectiveness. Investing in digital marketing and telemedicine platforms will boost patient engagement and improve drug adherence, directly affecting product success.